National –
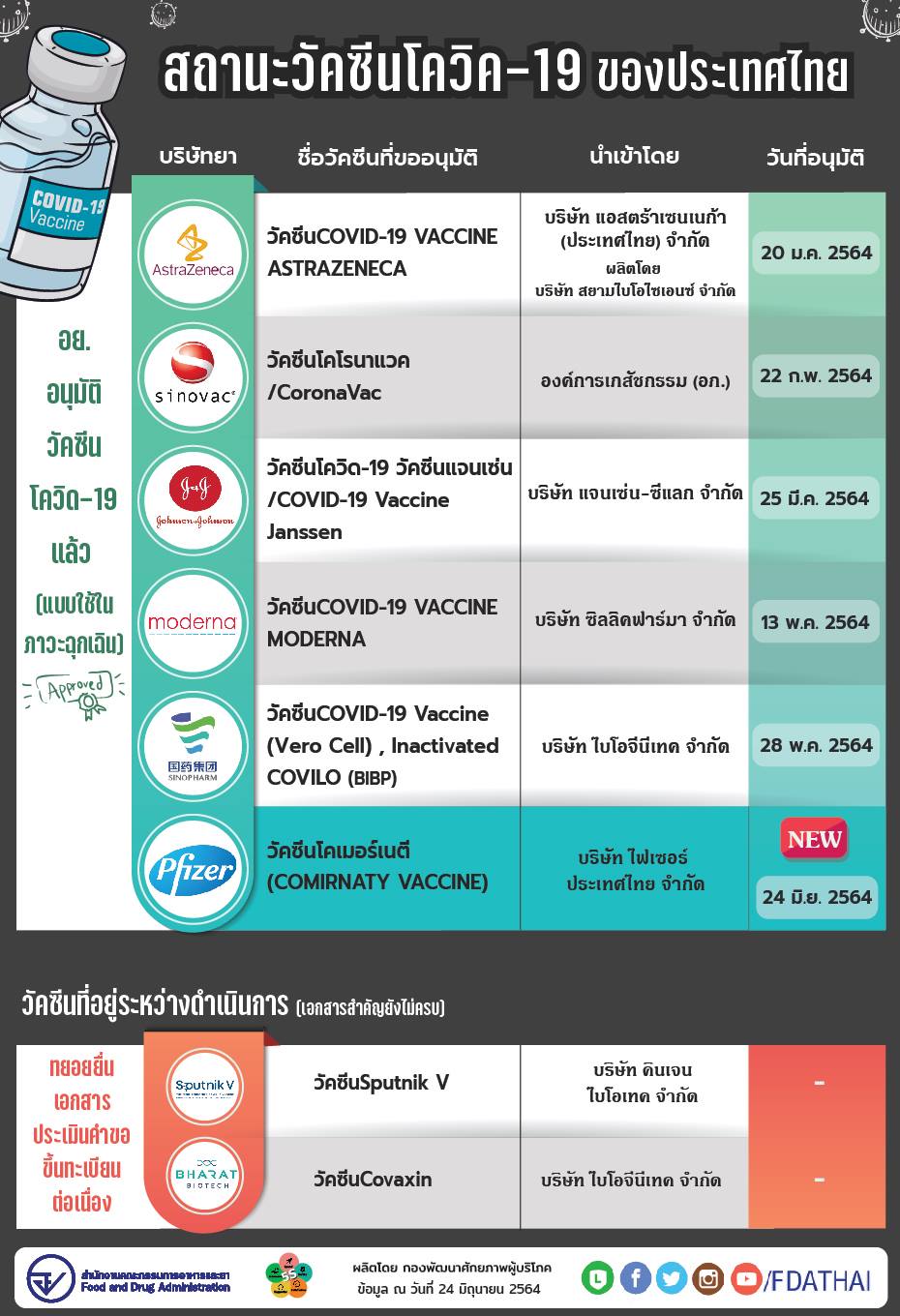
Thailand’s Food and Drug Administration (FDA) today, June 24th, has approved the registration of Pfizer Covid-19 vaccine for local emergency use, marking the sixth vaccine manufacturer to be officially distributed in Thailand.
Dr. Paisarn Dunkum, Deputy Secretary-General of the FDA, revealed to the Associated Press this afternoon that the FDA has approved the registration of the Comirnaty Vaccine, which is manufactured by Pfizer Co., Ltd and imported by Pfizer Thailand Co., Ltd. as Thailand’s sixth official Covid-19 vaccine for local distribution.
According to the schedule above, the FDA has already approved 5 Covid-19 vaccines include:
- AstraZeneca vaccine, imported by AstraZeneca (Thailand) Co., Ltd. and locally produced by Siam Bioscience Co., Ltd.
- Coronavac vaccine of Sinovac Life Sciences Co., Ltd., imported by the Government Pharmaceutical Organization (GPO)
- Johnson & Johnson Vaccine by Jansen Zelac Co., Ltd.
- Moderna vaccine, imported by Zuellig Pharma Co., Ltd.
- SinoFarm vaccines Imported by Biogenie Tech Co., Ltd.
Two additional brand vaccinations, Sputnik V and Covaxin are currently under the process of registration to the FDA.
Although now approved for emergency use, TPN media notes that doses of Pfizer are not expected to be available in Thailand in numerous quantities until around Quarter 4.
-=-=-=-=-=-=-=-=-=-=-=-=-=-=–=–=–=–==-
Help support The Pattaya News in 2021!
Follow us on Facebook, Twitter, Google News, Instagram, Tiktok, Youtube, Pinterest, Flipboard, Parler, or Tumblr
Join us on LINE for breaking alerts!
Join the discussion in our Facebook group https://www.facebook.com/groups/438849630140035/or in the comments below.
Want to advertise with us or inquire about promoting your business, event, charity, or sporting event? E-mail us at Pattayanewseditor@gmail.com
Have a General news tip, press release, question, comment, etc? We are not interested in SEO proposals. Mail us at Pattayanewseditor@gmail.com